Abstract: Calcium titanate (CaTiO₃), a classic perovskite oxide material, has emerged as a promising candidate for advanced energy applications, particularly in solar cells and photocatalysis. The development of high-quality CaTiO₃ sputtering targets enables precise thin film deposition through physical vapor deposition techniques, offering controlled stoichiometry, excellent crystallinity, and tunable optoelectronic properties. This article comprehensively reviews the synthesis of CaTiO₃ sputtering targets, thin film deposition methodologies, structural and optical properties, and applications in perovskite solar cells and photocatalytic systems.
Table of Contents
- What is Calcium Titanate (CaTiO₃) ?
- Synthesis and Fabrication of CaTiO₃ Sputtering Targets
- Thin Film Deposition Techniques
- Structural and Optical Properties
- Applications in Perovskite Solar Cells
- Photocatalytic Applications
- Challenges and Future Perspectives
- Conclusion
- References
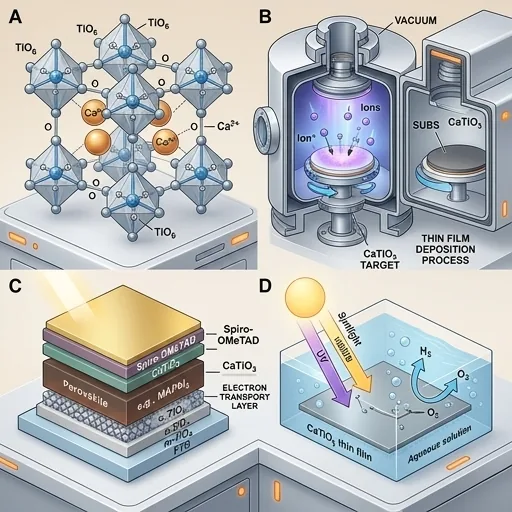
What is Calcium Titanate (CaTiO₃)?
Perovskite-structured materials have revolutionized the field of optoelectronics and energy conversion due to their exceptional electronic, optical, and catalytic properties. Among these, calcium titanate (CaTiO₃) represents a fundamental perovskite oxide with the general formula ABO₃, where Ca occupies the A-site and Ti occupies the B-site in an octahedral coordination [1].
The material exhibits orthorhombic symmetry (space group Pbnm) with lattice parameters:
- a = 5.3862 Å
- b = 5.4433 Å
- c = 7.6440 Å [2]
This structural versatility, combined with its favorable bandgap (~3.5 eV) and excellent chemical stability, makes CaTiO₃ an attractive material for thin film applications in energy conversion devices. The development of high-quality sputtering targets is crucial for reproducible thin film deposition, with magnetron sputtering offering advantages such as excellent film uniformity, controlled stoichiometry, and scalability for industrial applications.
Synthesis and Fabrication of CaTiO₃ Sputtering Targets
The quality of sputtering targets begins with the synthesis of high-purity, phase-pure CaTiO₃ powders, followed by densification processes to achieve targets suitable for physical vapor deposition.
Powder Synthesis Methods
Various synthesis methods have been developed, each offering distinct advantages for target fabrication:
Spray Pyrolysis Method
Lanfredi et al. demonstrated the synthesis of single-phase crystalline CaTiO₃ with spherical particle morphology using spray pyrolysis [2]. This method involves:
- Atomizing precursor solutions containing calcium nitrate and titanium isopropoxide
- Thermal decomposition in a furnace
- Production of nanometric to submicrometric particle sizes ideal for target fabrication
Microwave-Assisted Hydrothermal Synthesis
Moreira et al. reported rapid crystallization and controlled morphology using microwave-assisted hydrothermal methods [3]. This technique produces powders with specific structural characteristics, including:
- Random polycrystalline distortions
- Tilting between adjacent TiO₆ octahedra (influencing optical properties)
- High phase purity with minimal secondary phases
Solid-State Reaction
Conventional solid-state synthesis involves high-temperature calcination of calcium carbonate (CaCO₃) and titanium dioxide (TiO₂) mixtures. While straightforward, this method often requires:
- Multiple calcination steps (typically 1100–1200°C)
- Intermediate grinding to achieve phase purity
- Longer processing times compared to wet chemistry methods
Target Fabrication Process
The fabrication of CaTiO₃ sputtering targets typically involves several key steps:
- Powder Processing: Synthesized powders are subjected to ball milling to achieve uniform particle size distribution (typically 1–10 μm)
- Binder Addition: Organic binders such as polyvinyl alcohol (PVA) or polyethylene glycol (PEG) are added to improve green strength during pressing
- Cold Isostatic Pressing (CIP): The powder-binder mixture is pressed under high pressure (100–300 MPa) to form green compacts
- Sintering: Green compacts are sintered at 1200–1400°C in air or controlled atmospheres to achieve:
- High density (>95% theoretical density)
- Mechanical strength sufficient for sputtering
- Phase stability during plasma exposure
- Machining: Sintered targets are machined to precise dimensions and surface finish (typically Ra < 0.8 μm) suitable for sputtering applications
Critical Note: The quality of the sputtering target significantly influences thin film properties, including stoichiometry, crystallinity, and defect concentration. Target density and uniformity are paramount for preventing arcing and ensuring consistent deposition rates.

Thin Film Deposition Techniques
Reactive Magnetron Sputtering
Reactive magnetron sputtering is widely employed for CaTiO₃ thin film deposition, particularly for large-area applications. Ohtsu et al. investigated CaTiO₃ thin films deposited on titanium substrates using this method [1].
Key Process Parameters:
- Target: Dense CaTiO₃ ceramic target
- Substrate Temperature: 873 K (600°C)
- Oxygen Flow Rate: Varied from 1 to 10 sccm to control stoichiometry
Critical Findings:
- Films deposited with low oxygen flow exhibited Ti/Ca ratios lower than stoichiometric CaTiO₃ due to calcium hydroxide formation
- Increasing oxygen flow to 10 sccm resulted in films with Ti/Ca ratios nearly identical to stoichiometric CaTiO₃
- A titanium dioxide layer formed at the film-substrate interface, with thickness increasing with oxygen flow rate
- Deposited films crystallized into perovskite-type calcium titanate structure
Pulsed Laser Deposition (PLD)
Pulsed laser deposition offers advantages for stoichiometric transfer from target to substrate. In comparative studies, PLD has shown superior capability for depositing stoichiometric CaTiO₃ films without significant interfacial oxide layer formation [1].
Advantages of PLD:
- Excellent stoichiometric transfer from target to film
- Lower substrate temperature requirements compared to sputtering
- Reduced interfacial reaction layers
- Enhanced control over film crystallinity and orientation through laser fluence and pulse frequency control
Alternative Deposition Methods
While sputtering and PLD are primary techniques, other methods include:
- Metal-Organic Chemical Vapor Deposition (MOCVD): Offers conformal coverage and excellent thickness control but requires precise control of precursor chemistry and decomposition kinetics
- Sol-Gel Processing: Cost-effective for research-scale investigations but less suitable for industrial sputtering target applications requiring high-density coatings
Structural and Optical Properties
Crystal Structure and Phase Formation
CaTiO₃ crystallizes in the orthorhombic perovskite structure (space group Pbnm) at room temperature, transforming to tetragonal and cubic phases at elevated temperatures. The material exhibits a tolerance factor (t) ≈ 0.97, indicating slight distortion from ideal cubic perovskite structure.
Deposition Parameters Influencing Crystallinity:
| Parameter | Effect on Film Quality |
|---|---|
| Substrate Temperature | >600°C promotes better crystallinity and preferred (hkl) orientation |
| Oxygen Partial Pressure | Critical for maintaining stoichiometry; prevents oxygen vacancies |
| Post-Deposition Annealing | Often required at 700–800°C to improve crystallinity and reduce grain boundaries |
Optical Properties
CaTiO₃ exhibits a direct bandgap of approximately 3.5 eV, making it transparent in the visible region but active in the ultraviolet range. Moreira et al. established the relationship between structural tilting of TiO₆ octahedra and photoluminescence properties [3]:
- Structural distortions create intermediate energy states within the bandgap
- These states are responsible for photoluminescence emissions in the visible range
- Synthesis method influences the nature and concentration of these defect states
Bandgap Engineering: The optical properties can be tuned through doping and defect engineering. For instance, self-deficient CaTiO₃ has shown improved performance for solar cell applications by introducing mid-gap states [6].
Applications in Perovskite Solar Cells
Interface Modification Layer
CaTiO₃ has emerged as a promising interface modification material in perovskite solar cells (PSCs). Li et al. demonstrated that CaTiO₃ interface modification significantly improves photovoltaic performance [4]:
Key Benefits:
- Enhanced Electron Extraction: CaTiO₃ forms an effective electron transport layer (ETL) that facilitates efficient electron extraction from the perovskite active layer to the electrode
- Reduced Recombination: Acts as a passivation layer, reducing interfacial recombination losses and improving open-circuit voltage (VOC)
- Improved Stability: Protects the perovskite layer from moisture and oxygen ingress, enhancing device longevity
Performance Metrics: Devices incorporating CaTiO₃ interface layers showed improved power conversion efficiency (PCE), with enhancements attributed to better energy level alignment and reduced charge recombination at the ETL/perovskite interface.
Self-Deficient CaTiO₃ for Enhanced Performance
Recent research explores self-deficient CaTiO₃ for improved solar cell performance. Pandey et al. reported significant improvements in efficiency for new-generation solar cells [6]:
The controlled introduction of defects creates intermediate energy states that:
- Enhance light absorption in the visible region (extending beyond 3.5 eV limitation)
- Improve charge carrier separation and transport
- Reduce recombination rates through trap state management
- Enable better band alignment with adjacent organic or inorganic layers
Integration with Other Materials
CaTiO₃ has been successfully integrated with various photovoltaic architectures:
- Hybrid Perovskites: Effective ETL for MAPbI₃ and mixed-cation/halide perovskites
- Organic Photovoltaics: Promising interface material in organic-inorganic hybrid solar cells
- Tandem Structures: Explored in 2-terminal and 4-terminal tandem solar cell configurations as recombination layers or filtering layers
Photocatalytic Applications
Hydrogen Production
CaTiO₃ demonstrates excellent photocatalytic activity for hydrogen production through water splitting. Han et al. developed CdSe/CaTiO₃ nanocomposites showing remarkable enhancement in photocatalytic hydrogen production [5]:
Performance Highlights:
- 48× higher hydrogen production rate than pure CaTiO₃
- 4.14× higher performance than CdSe quantum dots alone
- Excellent stability over multiple photocatalytic cycles
Mechanism of Enhancement:
- Extended Light Absorption: CdSe quantum dots (QDs) extend light absorption into the visible region (550–700 nm)
- Type-II Heterojunction: Facilitates efficient separation of photogenerated electron-hole pairs
- Spatial Charge Separation: Electrons accumulate on CaTiO₃ conduction band while holes remain in CdSe valence band
Environmental Remediation
Organic Pollutant Degradation CaTiO₃ thin films effectively degrade organic pollutants (dyes, pesticides, pharmaceuticals) under UV irradiation through generation of reactive oxygen species (ROS), including hydroxyl radicals (·OH) and superoxide anions (O₂·⁻).
Antimicrobial Applications Photocatalytic coatings exhibit antimicrobial activity through oxidative stress mechanisms, making them suitable for:
- Self-cleaning surfaces in medical facilities
- Water purification systems
- Antifouling coatings for marine applications
CO₂ Reduction Recent studies explore CaTiO₃-based systems for photocatalytic reduction of CO₂ to valuable fuels (CO, CH₄, CH₃OH) using Z-scheme heterojunction architectures.
Heterojunction Engineering
Type-II Heterojunctions Combining CaTiO₃ with narrow bandgap semiconductors (CdS, CdSe, g-C₃N₄) creates staggered band alignments that improve charge separation and inhibit recombination.
Z-Scheme Systems CaTiO₃-based Z-scheme photocatalysts maintain strong redox capabilities by preserving high-energy electrons and holes in different components while allowing spatial separation.
Doped Systems Strategic doping with transition metals (Fe, Co, Ni) or non-metals (N, S) modifies the band structure, introducing defect states that enhance visible-light photocatalytic activity.
Challenges and Future Perspectives
Current Limitations
Despite promising results, several challenges remain in CaTiO₃-based device development:
- Bandgap Limitation: The wide bandgap (~3.5 eV) limits visible light absorption, requiring sensitization or doping strategies for solar energy applications
- Interface Optimization: Further optimization of CaTiO₃ interfaces with various substrates and active layers is needed to minimize lattice mismatch
- Scalability: Large-area deposition of high-quality CaTiO₃ thin films remains challenging for industrial roll-to-roll processing
- Stability Under Operation: Long-term stability under operational conditions (thermal cycling, humidity) requires further investigation, particularly for interface layers in PSCs
Future Research Directions
Advanced Doping Strategies Exploration of co-doping (acceptor + donor) and gradient doping profiles to optimize electronic properties while maintaining phase stability.
Nanostructured Films Development of nanostructured CaTiO₃ films (nanorods, nanosheets, mesoporous structures) with enhanced surface area and light-trapping capabilities for photocatalytic applications.
Machine Learning Optimization Application of machine learning algorithms for:
- Optimization of deposition parameters (pressure, temperature, gas flow)
- Prediction of doping effects on bandgap and conductivity
- High-throughput screening of heterojunction combinations
Integration with Emerging Technologies
- Photoelectrochemical cells for unassisted water splitting
- Memristive devices utilizing ionic conductivity of CaTiO₃
- Photodetectors exploiting the UV-sensitive optical properties
Conclusion
CaTiO₃ sputtering targets represent a critical enabling technology for the deposition of high-quality perovskite thin films for energy applications. Through controlled synthesis—utilizing spray pyrolysis, hydrothermal methods, or solid-state reactions—and optimized deposition parameters (reactive sputtering, PLD), CaTiO₃ thin films with tailored structural, optical, and electronic properties can be achieved.
The material demonstrates versatile applications:
- As interface modification layers in perovskite solar cells, enhancing efficiency and stability
- In photocatalytic systems for sustainable hydrogen production and environmental remediation
- As heterojunction components in advanced Z-scheme photocatalysts
Future advancements in target fabrication (higher density, larger diameters), deposition techniques (high-rate sputtering), and device engineering (multifunctional interfaces) will further enhance the performance and applicability of CaTiO₃-based systems. As research continues to address bandgap limitations and optimize interfacial properties, CaTiO₃ is poised to play an increasingly important role in next-generation energy conversion and storage technologies.
AtoZmat —— Advanced Materials from A to Z.
References
[1] Ohtsu, N.; Ito, A.; Saito, K.; Hanawa, T. Characterization of Calcium Titanate Thin Films Deposited on Titanium with Reactive Sputtering and Pulsed Laser Depositions. Surface and Coatings Technology 2007, 201 (18), 7686–7691. DOI: 10.1016/j.surfcoat.2007.02.043
[2] Lanfredi, S.; Storti, F.; Simões, L. P. M.; Djurado, E.; Nobre, M. A. L. Synthesis and Structural Characterization of Calcium Titanate by Spray Pyrolysis Method. Materials Letters 2017, 201, 148–151. DOI: 10.1016/j.matlet.2017.05.001
[3] Moreira, M. L.; Paris, E. C.; do Nascimento, G. S.; Longo, V. M.; Sambrano, J. R.; Mastelaro, V. R.; Bernardi, M. I. B.; Andrés, J.; Varela, J. A.; Longo, E. Structural and Optical Properties of CaTiO₃ Perovskite-Based Materials Obtained by Microwave-Assisted Hydrothermal Synthesis: An Experimental and Theoretical Insight. Acta Materialia 2009, 57 (17), 5174–5185. DOI: 10.1016/j.actamat.2009.07.019
[4] Li, S.; Li, X.; Yang, J.; Jiang, Q.; Lai, H.; Tan, Y.; Xiao, B.; Xu, T. Improvement of Photovoltaic Performance of Perovskite Solar Cells by Interface Modification with CaTiO₃. Journal of Power Sources 2020, 449, 227504. DOI: 10.1016/j.jpowsour.2019.227504
[5] Han, J.; Liu, Y.; Dai, F.; Zhao, R.; Wang, L. Fabrication of CdSe/CaTiO₃ Nanocomposites in Aqueous Solution for Improved Photocatalytic Hydrogen Production. Applied Surface Science 2018, 459, 520–526. DOI: 10.1016/j.apsusc.2018.08.026
[6] Pandey, S.; Shukla, A.; Tripathi, A. Improvement in the Performance and Efficiency on Self-Deficient CaTiO₃: Towards Sustainable and Affordable New-Generation Solar Cells. Materials Today Communications 2023, 35, 105575. DOI: 10.1016/j.mtcomm.2023.105575